Color anodizing is a popular process because it allows for the creation of products in a wide range of colors. Such an enhancement of appearance can make a product more appealing to a potential customer. Buyers of color anodized metals should keep in mind the limitations of the color anodizing process, though. Read More…
Alexandria Metal Finishers provides many finishing services, one of which is anodizing. Hardcoat anodizing, aluminum anodizing, hardcoat with Teflon® plus chromic and sulfuric anodizing are some of our processes. We provide services to aerospace, commercial, electronics, medical industries and more.
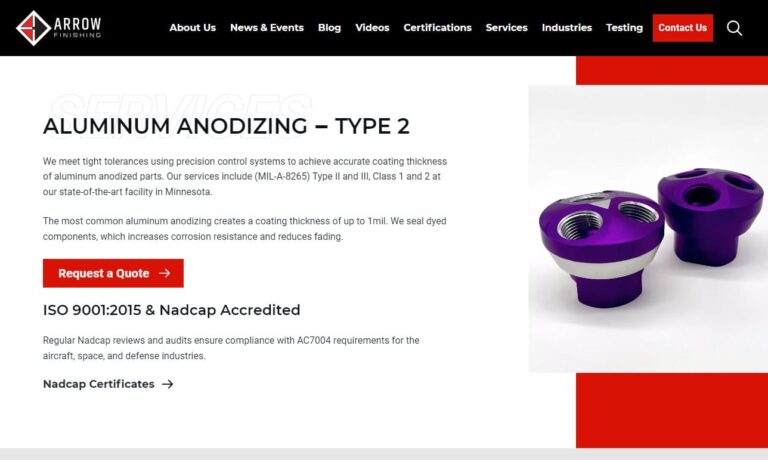
We specialize in aluminum anodizing, chromate conversion coatings, hard anodizing and sulfuric anodizing. To assure process repeatability and finish consistency, we use computer-controlled processing. All of our processes meet military specifications. Our quality system is certified to ISO 9001:2015, AS-9100 Rev D & NADCAP.
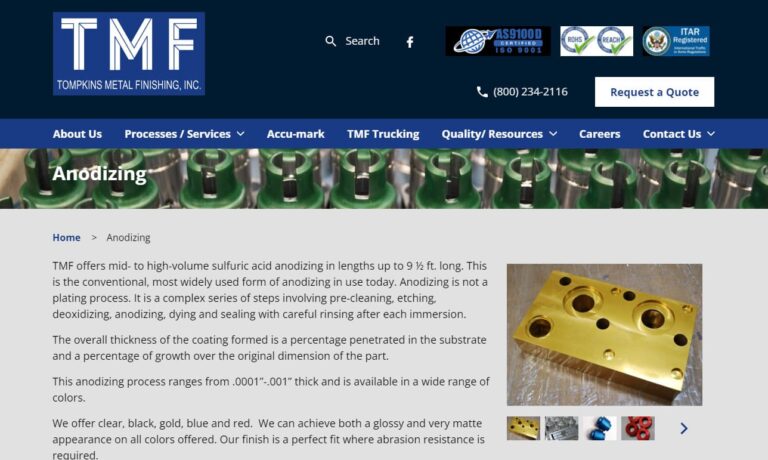
At Tompkins Metal Finishing, we offer mid to high volume aluminum anodizing. We do pre-cleaning, etching, deoxidizng, dyeing and sealing. We can achieve a wide range of surface finishes from bright to dull matte in clear, black, gold, blue and red. Other methods are also available with consideration of alloy, coating thickness, masking requirements and racking instructions.
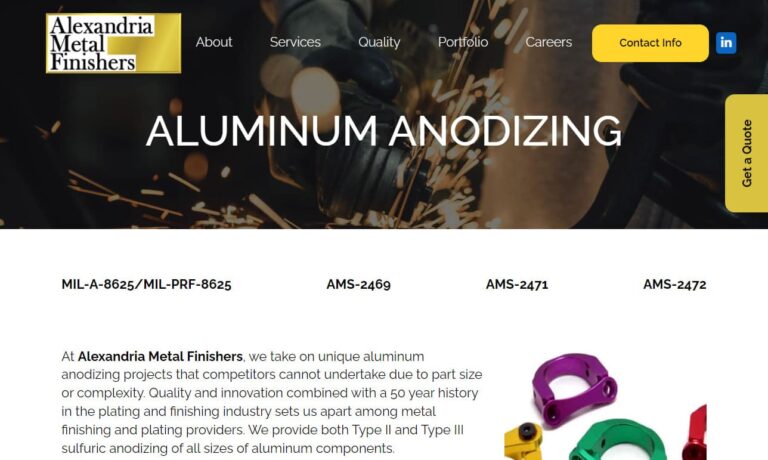
At Alexandria Metal Finishers, Inc., we are proud to offer comprehensive aluminum anodizing services tailored to the specific needs of our customers. We deliver high-quality anodized aluminum products that meet the highest standards of durability, aesthetics, and performance. Our aluminum anodizing process involves carefully preparing the surface of aluminum components and subjecting them to an...
When you choose INCERTEC, you're choosing a partner committed to excellence in every aspect of aluminum anodizing. Trust us to meet your specific requirements with a dedication to quality that goes beyond expectations. Reach out to us today for more information, and let us elevate your projects to new heights.
Dajcor Aluminum is the leading Canadian supplier of extruded, fabricated/machined and anodized components and assemblies to the automotive, renewable energy, transportation, building trades, military, recreation, and consumer-product industries.
More Color Anodizing Manufacturers
For example, some colors fade more easily than others. Black and gold dyes are made by inorganic means; namely they are the result of combining a chemical mixture called ferric ammonium oxalate to an anodized product. This inorganic dye tends to be more lightfast than other colors. Organic colors, which are much more varied, have the downside of being prone to fade. Metallic colors are limited to metallic hues, but remain bright longer then either of the first two processes.
Customers should consult professionals before making decisions about the color of their anodized products. What type of product is being produced will also play a part in what color a customer chooses. For example, the architecture industry tends to use mostly metallic color processes, while organic dyes, used in the electronic and construction fields work best with aluminum.
The actual dying techniques vary depending on the dye being used as well as the metal. Some metals do not require the additional step of dying to create a variety of colors. Anodized titanium naturally produces color, what hue depending on the amount of electrical current used to anodize the piece. Many metal accessories such as body piercing studs, wedding rings and costume jewelry are made from titanium.
However, the most commonly employed color anodization comes from organic and chemical dyes applied directly after the anodizing treatment. Because anodizing increases the pore size of the chemical body at the surface of a metal, it soaks up and retains color well. Once a metal has been dyed, hot water or steam, often mixed with nickel acetate, is used to seal the surface and to convert the oxide into its hydrated form.
This process decreases bleed out and can improve corrosion resistance. Whether the color is a result of the process of anodizing or added, it is just one more reason anodized products are in such high demand.






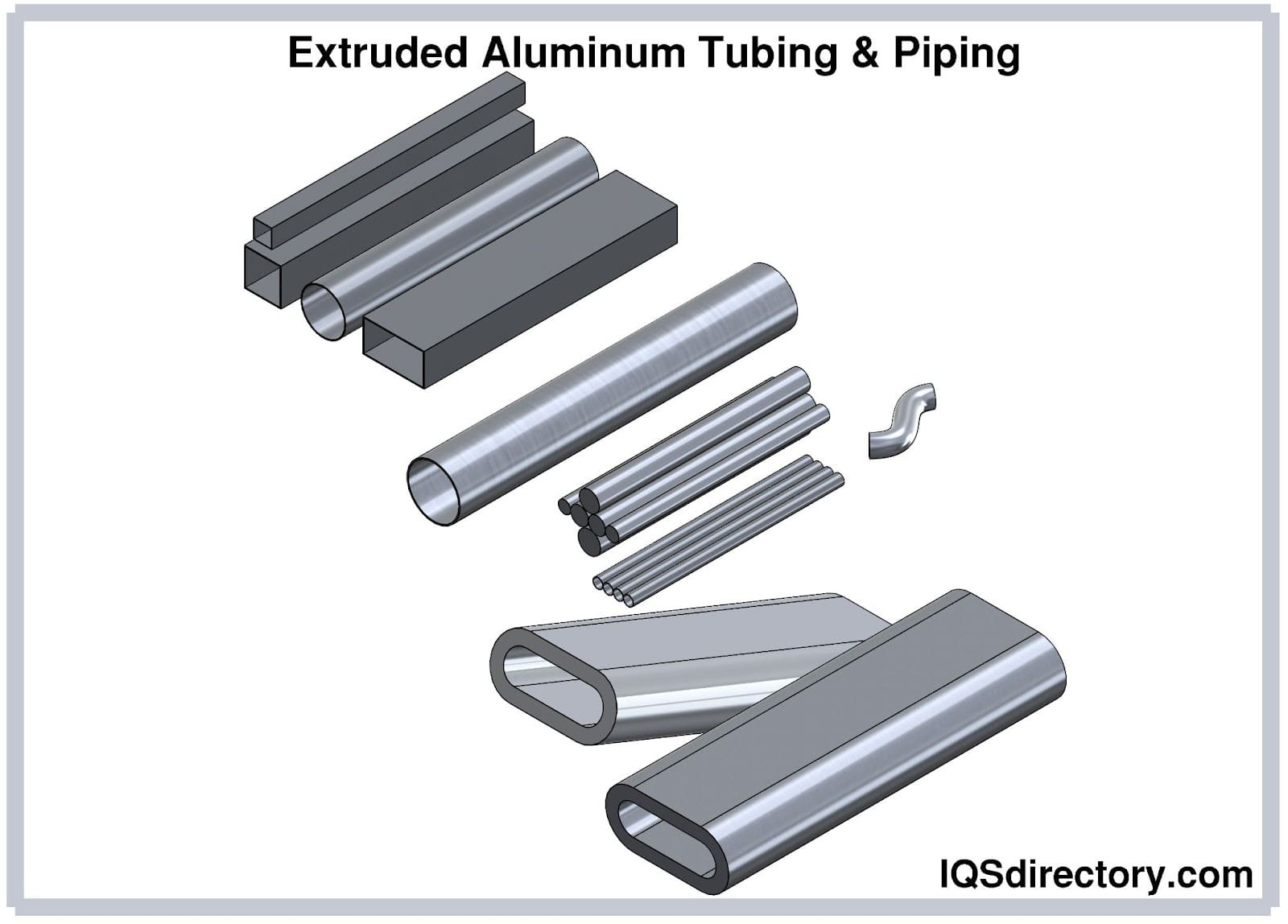



 Aluminum Anodizing
Aluminum Anodizing EDM
EDM Electroless Nickel Plating
Electroless Nickel Plating EMI Shielding
EMI Shielding Heat Treating
Heat Treating Metal Coating Services
Metal Coating Services Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services