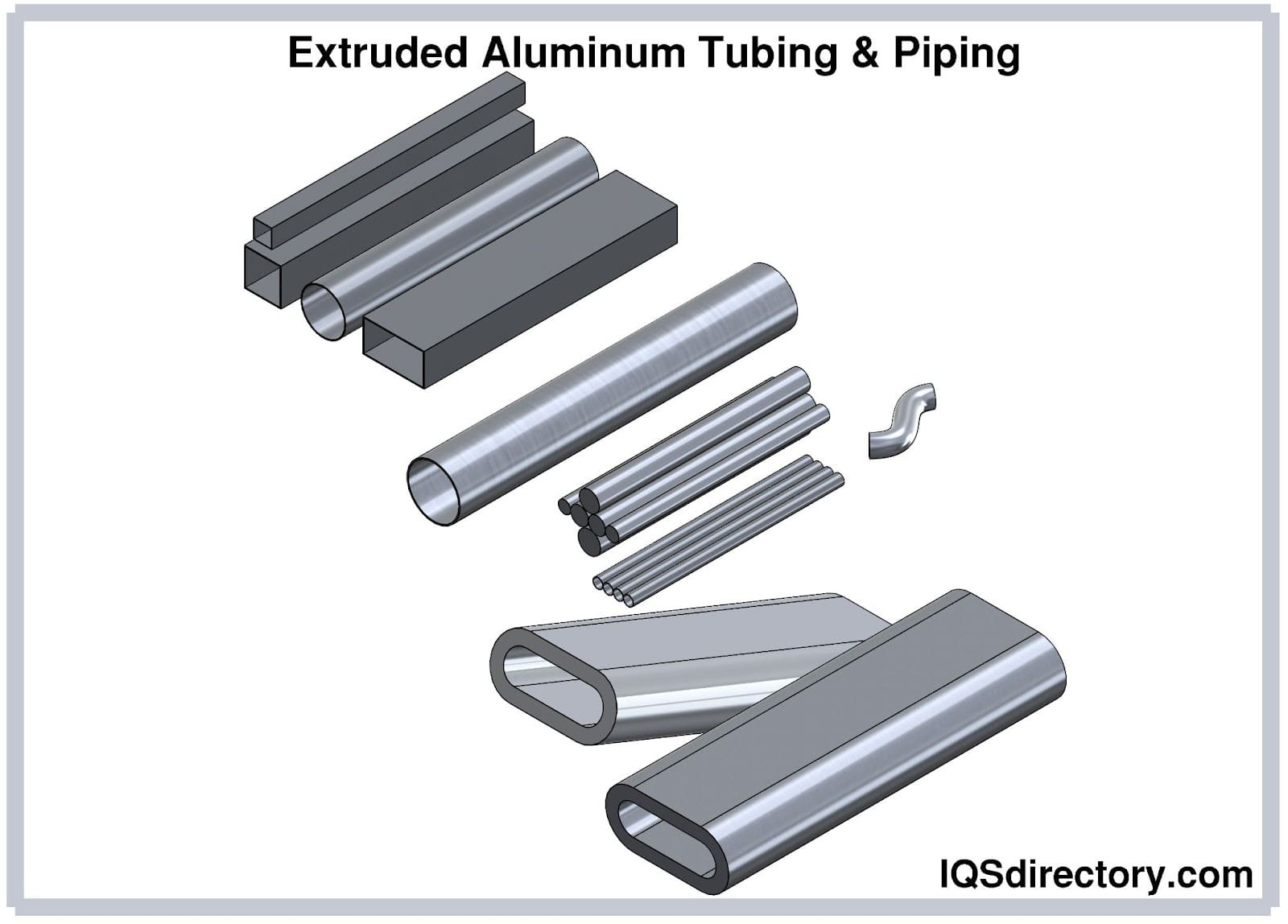
While anodizers most frequently work with aluminum alloys, due to the decreased corrosion resistance that results from the addition of the alloyed elements such as copper, iron and silicon, they also work with various other metals including niobium, tantalum, magnesium, zinc, titanium and tungsten. As a result of the variety of workable metals, anodizers are able to provide metal materials and parts to a wide range of industries including: industrial manufacturing, for anodized bars, rods, angles and profiles used in various processing applications. Read More…
Alexandria Metal Finishers provides many finishing services, one of which is anodizing. Hardcoat anodizing, aluminum anodizing, hardcoat with Teflon® plus chromic and sulfuric anodizing are some of our processes. We provide services to aerospace, commercial, electronics, medical industries and more.
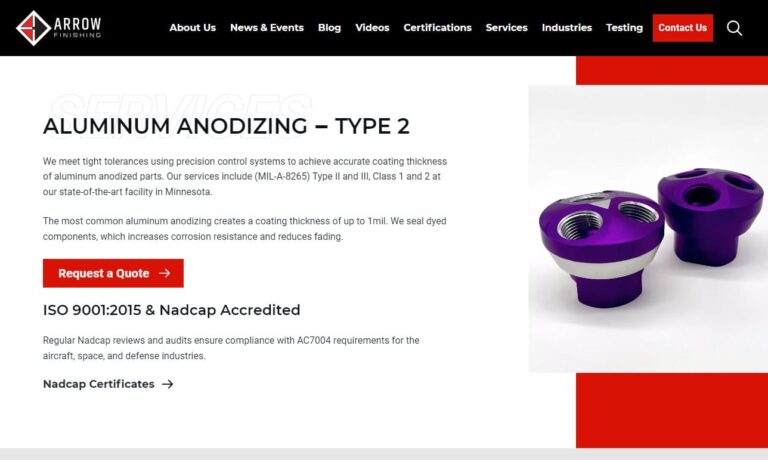
We specialize in aluminum anodizing, chromate conversion coatings, hard anodizing and sulfuric anodizing. To assure process repeatability and finish consistency, we use computer-controlled processing. All of our processes meet military specifications. Our quality system is certified to ISO 9001:2015, AS-9100 Rev D & NADCAP.
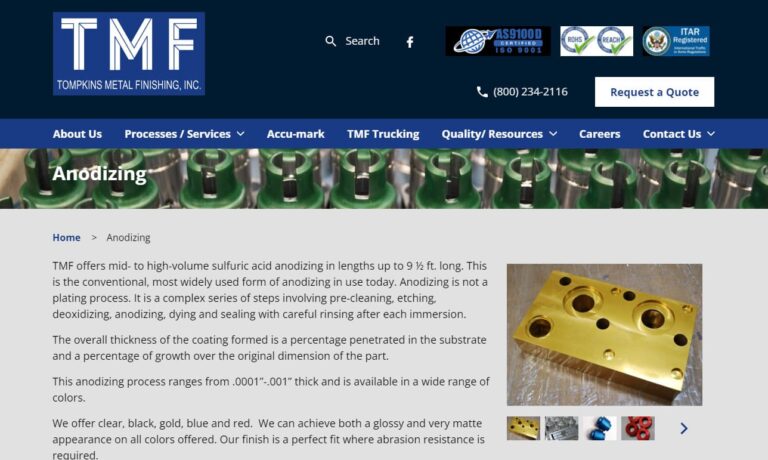
At Tompkins Metal Finishing, we offer mid to high volume aluminum anodizing. We do pre-cleaning, etching, deoxidizng, dyeing and sealing. We can achieve a wide range of surface finishes from bright to dull matte in clear, black, gold, blue and red. Other methods are also available with consideration of alloy, coating thickness, masking requirements and racking instructions.
At Alexandria Metal Finishers, Inc., we are proud to offer comprehensive aluminum anodizing services tailored to the specific needs of our customers. We deliver high-quality anodized aluminum products that meet the highest standards of durability, aesthetics, and performance. Our aluminum anodizing process involves carefully preparing the surface of aluminum components and subjecting them to an...
When you choose INCERTEC, you're choosing a partner committed to excellence in every aspect of aluminum anodizing. Trust us to meet your specific requirements with a dedication to quality that goes beyond expectations. Reach out to us today for more information, and let us elevate your projects to new heights.
Dajcor Aluminum is the leading Canadian supplier of extruded, fabricated/machined and anodized components and assemblies to the automotive, renewable energy, transportation, building trades, military, recreation, and consumer-product industries.
More Anodizer Manufacturers
These applications include electronics, for protective encasings of a variety of electrical equipment; automotive, for use as the trim and the body of the vehicle; military, for small parts such as fine aluminum wire as well as large military aerospace parts; and medical, for surgical instruments and implants, to name just a few.
In addition, several industries have certain standards that anodizers should ideally adhere to such as the American Architectural Manufacturers Association (AAMA) standard 611-98, for anodized architectural aluminum.
Anodizers can use several different anodizing processes, ranging from the common to the experimental and obscure. The main difference between many of these processes is the type of acidic solution that is used. The two main types of acids used are chromic acid and sulfuric acid. Chromic acid refers to the H2 CrO4, which is an oxidizing acid family that the Environmental Protection Agency has placed restrictions on. The uses of certain types of this acid have been classified as a human carcinogen.
While emissions resulting from sulfuric acid, which refers to a strong mineral acid that is very similar to chromic acid, are also restricted by the EPA, it is considered less dangerous than the emission of chromic acid. However, there are some new developments in chromic acid that are much more environmentally-friendly. Additional acidic solutions used by anodizers include citric acid, phosphoric acid, boric acid and organic acids.
The two main types of anodizing processes that anodizers use are bath anodizing and anodic painting, although bath anodizing is more common in industrial applications. In bath anodizing the metal is fully immersed in the acidic solution, while in anodic painting the metal ferrule of a paint brush is dipped in the acidic solution and then used to apply it to the metal. As a result, anodizers typically use bath anodizing for mass production and anodic painting for smaller projects.









 Aluminum Anodizing
Aluminum Anodizing EDM
EDM Electroless Nickel Plating
Electroless Nickel Plating EMI Shielding
EMI Shielding Heat Treating
Heat Treating Metal Coating Services
Metal Coating Services Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services