Anodized aluminum is, simply, aluminum that has undergone a process to create a highly-durable finish. To create anodized aluminum, the metal is immersed in a series of tanks where it undergoes an electrochemical process and an anodic layer is grown from the metal itself. Read More…
Alexandria Metal Finishers provides many finishing services, one of which is anodizing. Hardcoat anodizing, aluminum anodizing, hardcoat with Teflon® plus chromic and sulfuric anodizing are some of our processes. We provide services to aerospace, commercial, electronics, medical industries and more.
We specialize in aluminum anodizing, chromate conversion coatings, hard anodizing and sulfuric anodizing. To assure process repeatability and finish consistency, we use computer-controlled processing. All of our processes meet military specifications. Our quality system is certified to ISO 9001:2015, AS-9100 Rev D & NADCAP.
At Tompkins Metal Finishing, we offer mid to high volume aluminum anodizing. We do pre-cleaning, etching, deoxidizng, dyeing and sealing. We can achieve a wide range of surface finishes from bright to dull matte in clear, black, gold, blue and red. Other methods are also available with consideration of alloy, coating thickness, masking requirements and racking instructions.
At Alexandria Metal Finishers, Inc., we are proud to offer comprehensive aluminum anodizing services tailored to the specific needs of our customers. We deliver high-quality anodized aluminum products that meet the highest standards of durability, aesthetics, and performance. Our aluminum anodizing process involves carefully preparing the surface of aluminum components and subjecting them to an...
When you choose INCERTEC, you're choosing a partner committed to excellence in every aspect of aluminum anodizing. Trust us to meet your specific requirements with a dedication to quality that goes beyond expectations. Reach out to us today for more information, and let us elevate your projects to new heights.
Dajcor Aluminum is the leading Canadian supplier of extruded, fabricated/machined and anodized components and assemblies to the automotive, renewable energy, transportation, building trades, military, recreation, and consumer-product industries.
More Anodized Aluminum Manufacturers
What is Anodized Aluminum?
Anodized aluminum is, simply, aluminum that has undergone a process to create a highly-durable finish. To create anodized aluminum, the metal is immersed in a series of tanks where it undergoes an electrochemical process and an anodic layer is grown from the metal itself. Due to the fact that this anodized layer is created from the aluminum itself, rather than being painted on or applied, this anodized aluminum will never chip, flake, or peel, and, compared to other similar materials on the market, it is significantly more durable. In comparison to other metals like copper and stainless steel, anodized aluminum is 60% lighter and three times tougher than the raw material.

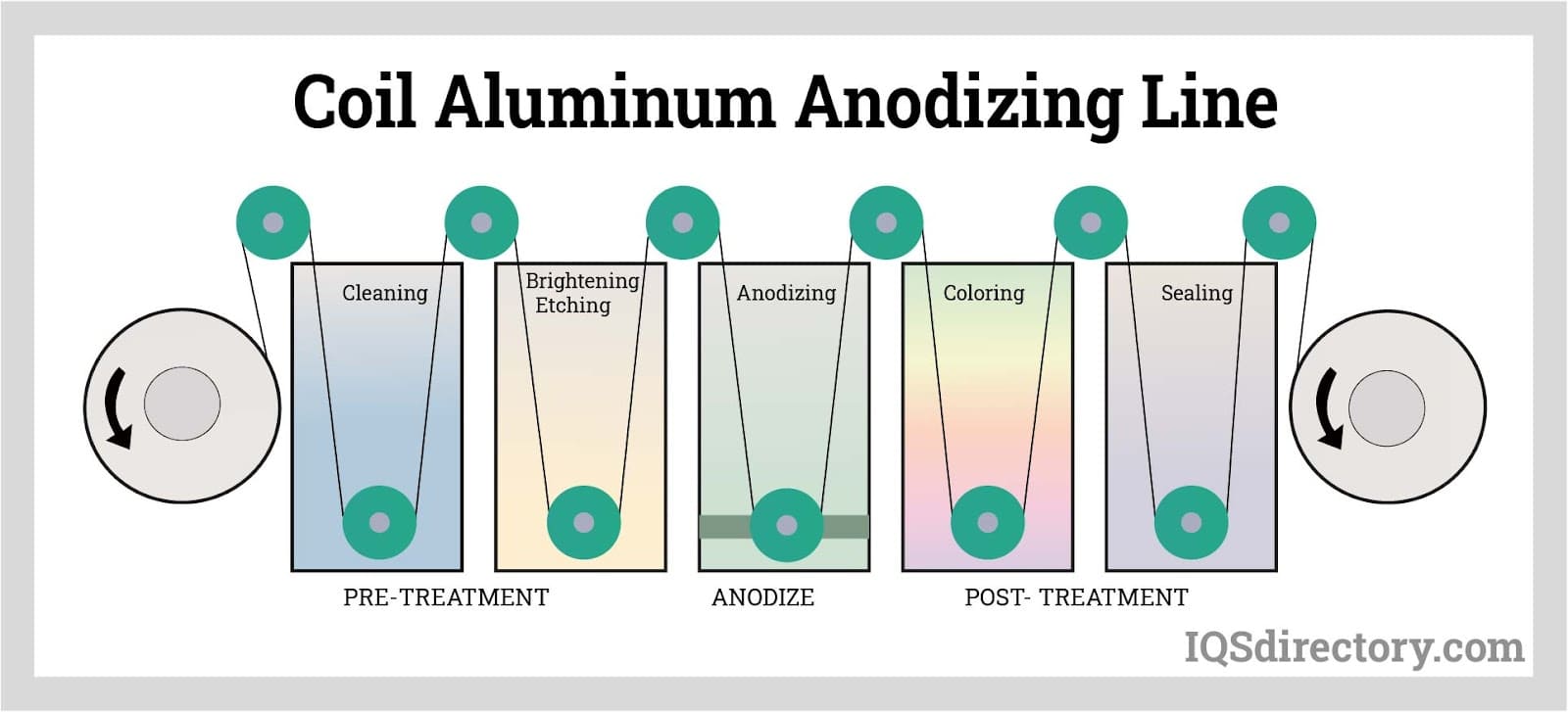
What Makes Anodized-Aluminum Coil Unique?
During the coil anodized-aluminum process, unwound coils of raw aluminum are pulled through a series of tanks where they are cleaned, colored, anodized, sealed, and then wound back up again. Anodized coil aluminum of the highest quality is created after this process is complete; it is strong, adaptable, and offers consistent color matching in accordance with the specific requirements of clients. Since the anodized metal is rolled, it is already prepared for transport.
A number of steps from the more-conventional, batch aluminum-anodizing process that increase costs and result in inconsistent finishes are removed in the coil anodized-aluminum process. An electrical contact point must be made during the batch aluminum-anodizing process in order for the electrical current to help the anodic layer expand. This is accomplished by fastening each item to be anodized with a metal hook, or rack, which frequently leaves burn marks on the material that must be trimmed off, increasing waste and cost.
What are the Benefits of Anodized Aluminum?
Durability
Aluminum is already a tough substance, but after the anodization procedure, the surface gets even stronger than the original aluminum. Once anodized, this material develops a surface that is three times tougher than regular aluminum and will not chip, flake, or peel, even after being colored. This product won't ever rust, tarnish, or weather because the anodization process is controlled by aluminum oxidation. Anodized aluminum is one of the strongest and most useful metal goods on the market.
Custom Color Options
Due to the open pore structure of the anodic layer before sealing, anodized aluminum can be processed to match almost any brand color or Pantone® swatch. Due to anodized aluminum’s adaptability, it can also be manufactured to mimic the appearance of other metals such as gold, bronze, copper, stainless steel, brass, and more without the worry of deterioration from the elements. There are various weathered-appearance finish options to pick from, though, if a worn-in appearance is required. Again, due to the properties of anodized aluminum, the color selected will not chip, peel, or flake over the course of the product's lifespan. Additionally, the UV-stable colors generated are guaranteed to last a long time.

Light in Weight
A lightweight metal, anodized aluminum weighs around 60% less than stainless steel, copper, or brass. This greatly reduces the cost of shipping this material that can be employed to address a variety of design problems. Anodized aluminum provides a flexible alternative whenever a light, strong, and appealing material is required, from architecture to consumer product design.
Where is Anodized Aluminum Used?
Any application that currently uses another metal material can use anodized aluminum. Manufactured anodized aluminum serves the following sectors:
Architecture
SInce aluminum does not rust, anodized aluminum is used for a variety of outdoor purposes like the framework for outdoor furniture, roofing systems, and storefront exteriors. It also provides an interesting exterior option for stunning, contemporary building rusts, patinas, or weathered effects. Anodized aluminum is a beautiful, lightweight, and cost-effective solution for interior finishes like ceiling panels, retail signage, and elevator panels because it can be manufactured to order in any color, finish, or texture.
Transportation
Aluminum that has been anodized is three times as durable as aluminum that has not been anodized. Anodized aluminum gives the durability and weather resistance vehicles need, as well as the great elegance customers demand. This is true of the interiors of commercial aircraft, rail car panels, and the brilliant, external trim of sports automobiles.
Consumer Goods
Customers seek out durable goods that are aesthetically pleasing and fit their way of life. Any appearance, texture, or color can be achieved with anodized aluminum. Anodized aluminum gives the adaptability and quality that your customers want in microwaves, coffee makers, and other consumer goods. Anodized aluminum can be a stunning component of any project or product, whether you're making eye-catching skyscrapers and structures, artwork, the newest line of luxury vehicles, or high-end appliances. The environmentally-friendly anodizing technique results in a finished material with unmatched and dynamic beauty, longevity, and durability.
Choosing the Correct Anodized Aluminum Supplier
To make sure you have the most productive outcome when purchasing Anodized Aluminum from an Anodized Aluminum Supplier, it is important to compare at least 4 Companies using our list of Anodized Aluminum manufacturers. Each Anodized Aluminum Manufacturer has a business profile page that highlights their areas of experience and capabilities and a contact form to directly communicate with the manufacturer for more information or request a quote. Review each Anodized Aluminum company website using our proprietary website previewer to get an idea of what each business specializes in, and then use our simple RFQ form to contact multiple Anodized Aluminum companies with the same quote.









 Aluminum Anodizing
Aluminum Anodizing EDM
EDM Electroless Nickel Plating
Electroless Nickel Plating EMI Shielding
EMI Shielding Heat Treating
Heat Treating Metal Coating Services
Metal Coating Services Castings & Forgings
Castings & Forgings Bulk Material Handling
Bulk Material Handling Electrical & Electronic Components
Electrical & Electronic Components Flow Instrumentation
Flow Instrumentation Hardware
Hardware Material Handling Equipment
Material Handling Equipment Metal Cutting Services
Metal Cutting Services Metal Forming Services
Metal Forming Services Metal Suppliers
Metal Suppliers Motion Control Products
Motion Control Products Plant & Facility Equipment
Plant & Facility Equipment Plant & Facility Supplies
Plant & Facility Supplies Plastic Molding Processes
Plastic Molding Processes Pumps & Valves
Pumps & Valves Recycling Equipment
Recycling Equipment Rubber Products & Services
Rubber Products & Services